
What causes bread dough to rise?
15 January 2021 | Gary Tucker, Fellow
A great deal is written about the way that yeast produces carbon dioxide to leaven bread dough during proof. However, there are other gases such as ethanol and steam that are vital during the oven stages of bread making. These gases tend to be forgotten but without them it would not be possible to bake bread of the quality we expect. Their role increases in importance as the bread reaches the end of the bake time when its structure becomes more susceptible to collapse. In this article I’ll discuss the different gases that are essential in helping bread dough to rise during baking.
There are many ways of making bread from the basic ingredients of flour, water, yeast and salt, from labour-intensive artisan bread to high-throughput sandwich bread. However, there are three aspects that are critically important to any type of bread making, and it all starts in the mixer:
- The mixing action entrains numerous air bubbles into the dough that can be inflated during proof and baking.
- Dough development takes place during the mixing process to convert the flour proteins into a gluten matrix that has the elasticity to allow the bubbles to expand in later stages.
- Gases are generated by yeast action and/or chemical leavening agents to expand the bubbles and create the desired aerated bread structure.
The role of gases in expanding the bubbles during proving and baking is fascinating but usually focuses on yeast converting sugars to carbon dioxide (CO2). Yeast metabolism in dough is complex and involves a short aerobic fermentation stage where oxygen from the bubbles is used up followed by a much lengthier anaerobic stage. The details of this are explained later. The mechanisms help to explain why the dough volume initially shrinks, why there is a lag before the dough gradually increases in volume and how oven spring takes place. However, as mentioned there are other leavening gases that play a vital role, particularly during baking, and without them the bread is certain to collapse in the oven. This article highlights the role of the other gases together with the mechanisms by which those gases work to inflate the bubbles.
Background research
Among the many research papers on bread dough leavening there a couple that are worthy of mention. These introduced the concept that gases other than carbon dioxide played a key role in expanding gas bubbles in dough. Moore and Hoseney (1985) calculated the carbon dioxide volume from expansion of the gas bubbles and concluded that carbon dioxide alone did not explain the increase in volume from dough to bread during baking. They thought that ethanol was significant, particularly around 70°C when its vaporisation rate increased rapidly. A further publication on this subject was by Bloksma (1990) who compared the Moore and Hoseney calculations with his own on partial pressures of gases during baking. Bloksma included carbon dioxide, ethanol and water in his own calculations of thermal expansion. He concluded that water (in the form of steam) contributed more than half of the oven spring volume, with ethanol and carbon dioxide responsible for much of the rest. These calculations were made up to 70°C because this was the stage when Bloksma considered that oven spring ended and further volume increase was negligible.
Gases involved in bread making
As mentioned previously, during bread baking there are several gases that contribute to the leavening of bread dough. Carbon dioxide is the main gas associated with yeast leavened bread, however, other gases that play a role are ethanol, nitrogen and steam. There is also a small contribution from low molecular weight volatile compounds formed during fermentation. The main gases and their roles are described in Table 1.
| Gas | Description |
|---|---|
| Carbon dioxide (CO2) | Generated by yeast during aerobic and anaerobic fermentation of glucose. Amylases convert starches in the flour to maltose, which is further converted to glucose by maltase from the yeast. Anaerobic fermentation is the dominant effect. |
| Ethanol (C2H5OH) | Generated by yeast during anaerobic fermentation of glucose. Molar quantities of ethanol are the same as for CO2 during anaerobic fermentation. Ethanol boils at 78.4°C so its influence on dough expansion is significant as the dough temperature approaches 70°C. |
| Water (H2O) | Approximately 40-60g is lost from a 900g dough piece (to make an 800g lidded loaf) during baking. Much of this will be water which will turn to steam late in the oven. |
| Nitrogen (N2) | Nitrogen is left in the bubbles when oxygen is used by both yeast and ascorbic acid. Nitrogen expands as it increases in temperature. Dissolved gases (oxygen and nitrogen) will also come out of solution as the dough liquid increases in temperature. |
| Other volatiles | Volatile molecules are also generated from fermentation, such as carboxylic acids, aldehydes, ketones and alcohols. |
Table 1: Gases released from bread during baking
Carbon dioxide (CO2)
Carbon dioxide generation takes place during two stages of fermentation because yeast can metabolise both aerobically and anaerobically. Aerobic fermentation is the first pathway and will continue until all the oxygen is used up and the conditions in the dough become anaerobic. There is competition with ascorbic acid for oxygen, added to increase gluten oxidation during mixing, and this limits the extent of aerobic fermentation. The yeast first metabolises glucose and oxygen to carbon dioxide and water, as in Equation 1. Glucose is generated by enzymic pathways from the starch in the flour to maltose and then to glucose. Oxygen comes from the air in the bubbles entrained during mixing.
C6H12O6 + 6O2 → 6CO2 + 6H2O Equation 1
Having used the available oxygen, subsequent fermentation during proof takes place with the dough in an anaerobic condition. Yeast obtains the oxygen needed directly from the glucose, according to Equation 2. This is by far the most dominant stage in yeast fermentation. Equal molar quantities of carbon dioxide and ethanol are produced from glucose breakdown, which are significant, and discussed later.
C6H12O6 → 2CO2 + 2C2H5OH Equation 2
It is important to note that the carbon dioxide produced from either fermentation mode does not go straight into the gas bubbles. At proof temperatures of 32-38°C, carbon dioxide is soluble in water to a level of about 1.0 g/kg. It first dissolves into the aqueous phase that surrounds the yeast cells and continues to dissolve until the liquid becomes saturated. During the aerobic stage, oxygen is used from the air bubbles and this reduces the pressure inside each bubble. They first shrink by about 20% as they now only contain nitrogen. Only when the water is saturated with carbon dioxide can it enter the bubbles to inflate them. Carbon dioxide can also dissolve in organic solvents such as the vegetable oil used in bread making. This helps in a small way to increase the quantity of dissolved carbon dioxide in the dough.
Ethanol (C2H5OH)
Equation 2 shows that ethanol and carbon dioxide are produced in equal molar quantities during anaerobic fermentation. Ethanol is readily soluble in water and organic solvents, to the extent that all the ethanol produced during fermentation dissolves into the liquid phases surrounding the gas bubbles. It boils at 78.4°C and is likely to have left from the bread soon after its boiling point of 78.4°C is reached. The rate of ethanol evaporation is likely to be at its highest when the internal core temperature is above 70°C.
The role of ethanol in dough behaviour is complex and is a subject for further investigation. Ethanol is known as a universal solvent because it allows both polar and non-polar compounds to dissolve. It therefore increases the solubility of carbon dioxide so that more can dissolve and be available to release from solution as the dough temperature rises during baking. This contributes to the sudden oven spring that happens early in the baking process. Ethanol also increases the solubility of the gliadin wheat protein fractions, which may have an impact on the rheological behaviour of dough because the gliadins are thought to confer extensibility to dough. Another property of ethanol is the role it plays in softening bread crumb.
Steam (H20)
Water boils at 100°C and so will be released later into the baking process than ethanol (BP 78.4°C) or carbon dioxide (BP -56.6°C). By the time steam is released into the bubbles it is likely that the structure of a baked product will have increased in ‘viscosity’ substantially because of starch gelatinisation that takes place above 60°C. However, the thin gas bubble walls will still contain a lot of water and are very soft at this stage. Without the increase in pressure from ethanol and steam it is likely that the delicate bubbles will collapse.
Nitrogen (N2)
Air contains approximately 20% oxygen and 80% nitrogen. Oxygen is used up by the yeast during its aerobic phase and is replaced in the gas bubbles by carbon dioxide once the liquid surrounding the gas bubbles is saturated. Nitrogen is unaffected by yeast fermentation and will expand as the bread temperature rises. The contribution of nitrogen to bubble expansion is less significant than with the gases that come out of solution. Nitrogen expansion is uniform with temperature compared with the instant volume increase when a gas comes out of solution.
Each gas exerts its pressure influence at a different stage in the oven, and without these gases working in series it is not possible to create a crumb structure based on such a delicate network of bubbles.
When are the gases released?
This is another interesting question. Soluble gases such as carbon dioxide and ethanol dissolve into the aqueous and organic liquids surrounding the bubbles. The extent of their solubility depends on pressure and temperature. As the dough temperature increases, the gases are forced out of solution and inflate the gas bubbles. If a bubble breaks and coalesces with an adjacent bubble, the gas is retained within the larger bubble and is not lost. Only a small amount of gas is lost from the top surface of the dough. Experiments have shown that this is not a significant quantity and only occurs towards the end of proof and more so with less well developed doughs.
Oven spring takes place quite early into the baking process. This is when a sudden increase in pressure occurs inside the bubbles, which causes the dough to force upwards rapidly. Expansion slows down when the sides and top surfaces dry out, set hard and they resist the upward forces. Further volume increase is limited and, with pan bread, causes a break in the side crusts towards the upper edges. This pressure increase is caused mostly by carbon dioxide coming out of solution but there is some contribution from nitrogen expansion and ethanol. Oven spring is usually completed in the first third of the bake.
The internal network of bubbles in the bread crumb remains delicate for the duration of the bake. Without a positive pressure to maintain them, the bubble walls are soft enough so they can easily collapse. The bubble pressure is first maintained by carbon dioxide because it boils at the lowest temperature (-56.6°C), followed by ethanol (78.4°C) and finally by water (100°C). Nitrogen expansion takes place gradually as the dough temperature increases throughout. Each gas exerts its pressure influence at a different stage in the oven, and without these gases working in series it is not possible to create a crumb structure based on such a delicate network of bubbles. Figure 1 illustrates the temperature rise in an 800g loaf of sandwich bread during baking, with the approximate regions in which the gases have their influence on maintaining the bubble pressure. Figure 2 is a micro CT (computerised tomography) image taken for a 2cm cube from a white sandwich bread that shows the fine gas bubble structure. It would not be possible to create such fine and delicate crumb structures without a series of gases to maintain the internal gas pressures.
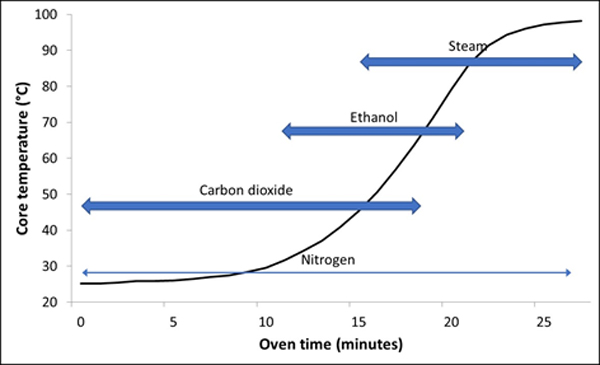
Fig 1: Time of influence of the different gases during baking of white sandwich bread

Fig 2: Micro CT image of the porous structure in a 2cm cube cut from of a white lidded loaf
Foam to sponge conversion
There is one last change in the bread crumb structure that is essential to avoid collapse of a loaf after baking. This is particularly important with bread baked in lidded pans where crust thickness and strength are much less than with open baked crusty products. Every single gas bubble must break to allow the hot gases out. If the intact gas bubbles cool, the result will be a loss in pressure and enough volume reduction to cause the side walls to collapse inwards. This change – where the bubbles break to allow the escape of their gases - is known as the foam to sponge conversion and is thought to happen just a few minutes before the bread exits the oven. The result is effectively one very complex gas bubble made from all the interconnected smaller gas bubbles.
Towards the end of the oven process, and just before the foam to sponge conversion, the gas bubbles contain a mixture of gases including nitrogen, steam, carbon dioxide, ethanol and some volatile organic compounds. It is likely that steam is mostly responsible for breaking each of the gas bubbles in bread; converting the structure from a closed foam to an open sponge. When water changes into a gas it increases in volume substantially. The later stages in baking are when the bread is hot enough for steam generation to occur rapidly.
This conversion is an essential part of the baking process and enables the gases to escape to atmosphere and be replaced by cooler air. Without this escape pathway each bubble suffers a massive collapse in pressure and volume as the gases cool.
Summary
The role of carbon dioxide, ethanol, nitrogen and steam is fascinating during mixing, proof and baking. It would not be possible to bake bread with a fine network of gas bubbles without each of these gases working together to maintain the pressure inside the bubbles. Carbon dioxide is responsible for the volume increase in dough during proof and for much of the oven spring that happens early into the bake. Ethanol and steam are vital to maintain pressure inside the delicate bubbles towards the later stages of baking. Finally, and after the starches have gelatinised and started to set, steam creates pressure that breaks each of the bubble walls to release the gases into the oven atmosphere. Air rushes back into the newly created single interconnected bubble to make the bread structure more resilient to collapse. By understanding these different mechanisms during mixing, proof and baking it is possible to design recipes and processes, and to solve baking problems more easily.
Understand baked products better with our baking courses
Understand baked products better with our baking courses from baking basics to biscuits and beyond. Take a look at the full range of high quality training that’s delivered by our experts to develop and grow your workforce‘s skills and talents.
How can we help you?
Do you need help solving your baking problems? Contact us to find out how we can help.







