High pressure processing of Innocent vegetable juices
Opportunity
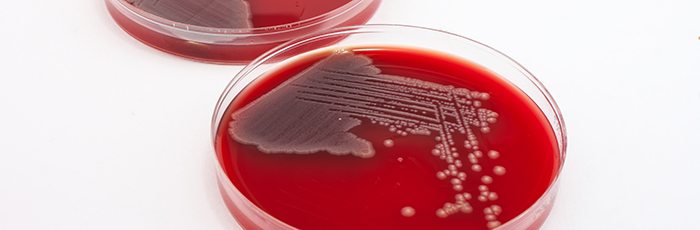
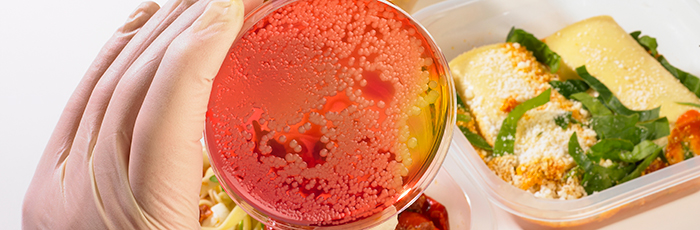
Innocent Drinks partnered with us for support in investigating how high pressure processing (HPP) could be applied to their vegetable juices to improve quality whilst maintaining safety. They wanted to determine if HPP would be sufficient to destroy the types of microorganisms that can limit the shelf-life of this type of product, and if any microorganisms surviving after processing would be able to grow within the intended storage conditions and shelf-life.
Download the complete Maximising Shelf-life eBook, for free, to unlock the tools for measuring and maximising the shelf-life of your food and drink products whilst ensuring their safety and quality.

Approach
Our team were able to advise as to the target microorganisms to assess and on appropriate packaging, treatment conditions and potential effects on the product, as well as assist in new product development.
For the analysis, we made use of our pilot HPP plant that is situated in our class 2 microbiology laboratory (where challenge tests can be performed on certain pathogens).
Results
As a result of our expertise and facilities, we were able to provide Innocent Drinks with the knowledge that the HPP processing was effective and that the products were microbiologically stable.
The Campden BRI difference
The food and drink industry presents a fast-paced, constantly changing environment that requires creative thinking, quick responses and cost-effective solutions to meet consumer demand. We can help overcome these challenges and support you in maximising shelf-life to get the most out of your products. Using state-of-the-art facilities and the latest methods and technology, you can be sure of a high-quality service that will help you to improve your products, processes and profitability, whilst protecting product safety and quality.
We provide many members and clients with tailored, expert support in all areas of shelf-life evaluation, maximisation and extension, and have specific expertise in microbiology, chemistry, sensory, regulatory and product innovation services. Get in touch to join those already benefitting from support.
Download our FREE Maximising Shelf-life eBook today!
Explore different strategies to maximise and extend product shelf-life, without compromising safety or quality.
How can we help you?
If you’d like to find out more about how we can help with the self-life of your products, contact our Support Team.